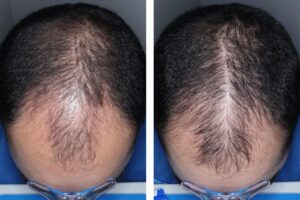
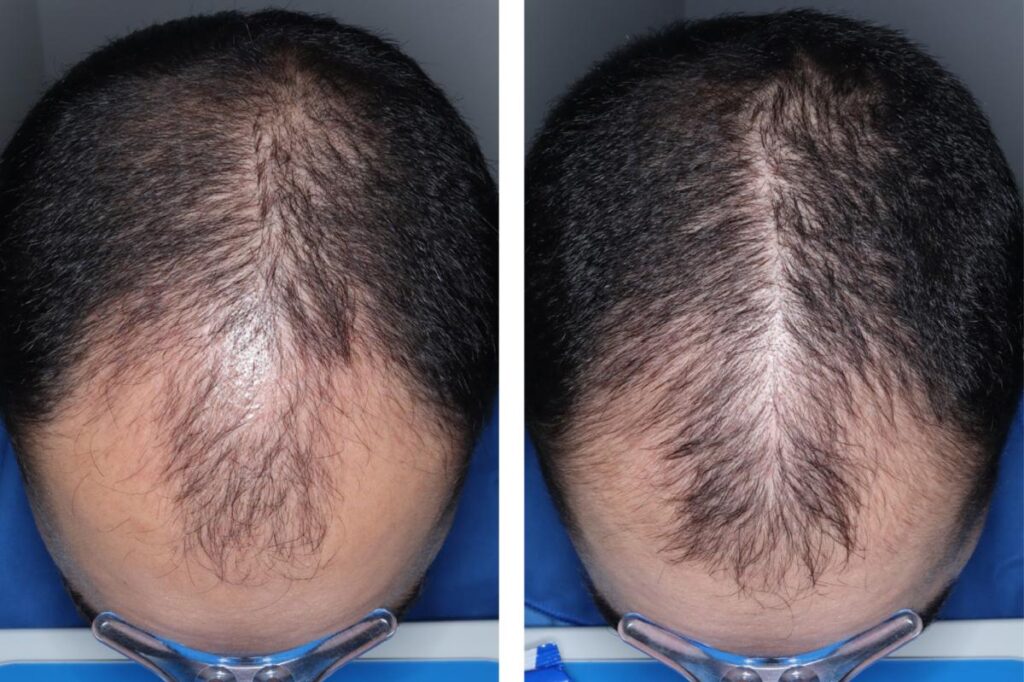
It’s not something to brush off.
Hair loss is a widespread problem in the US, with roughly 50 million men and 30 million women experiencing pattern baldness. No special potion completely reverses genetic or age-related decline, but there are a few products that seem to halt further loss, thicken existing hair and promote regrowth.
Now, a Connecticut-based biopharmaceutical company is sharing promising results of its experimental treatment, which is a revved-up spin on the active ingredient in Rogaine.
Men in a clinical trial grew an average of 30 to 33 hairs per square centimeter of their scalp in the six months after starting the minoxidil pill. Those on a placebo only had seven more hairs per square centimeter over the same period, Veradermics said.
“The positive data just announced [last week] … showed early, consistent and robust hair growth and demonstrated a favorable safety and tolerability profile with overall adverse event rates similar to placebo,” Dr. Reid Waldman, a Connecticut-based dermatologist and CEO of Veradermics, told The Post.
Veradermics hopes this medicine becomes the first FDA-approved non-hormonal oral treatment for pattern hair loss.
How the pill works
The formulation — known as VDPHL01 — contains nearly twice as much minoxidil (8.5 milligrams) as the maximum recommended daily dose (5 milligrams).
Minoxidil widens blood vessels to increase nutrient, oxygen and blood flow to budding hair follicles, extending their active growth phase while stimulating dormant follicles to become active.
Some 79% to 86% of trial participants on the pill reported “any improvement” in their hair growth after six months compared to 35% on the placebo.
Veradermics is testing a once-daily and a twice-daily dosing regimen in its clinical trials and hasn’t settled on the more favorable treatment.
Inside potential side effects
Oral minoxidil — more so than the topical version — carries serious heart risks, like a fast heart rate, fluid around the heart and chest pain.
No treatment-related cardiac issues were reported in this latest trial, which involved 519 participants with mild to moderate pattern hair loss.
What sets VDPHL01 apart from other minoxidil formulations, Waldman said, is that it provides “sustained and consistent exposures of minoxidil above the levels at which the drug has been observed to stimulate hair growth, while staying below the levels at which the drug has exhibited cardiac effects.”
Still, data in the most recent trial was only collected through six months. Veradermics plans to learn more about long-term use as trials progress.
Other side effects were reported. Nearly 6% of patients who took VDPHL01 experienced peripheral edema, fluid accumulation that causes swelling commonly in the hands, legs or feet.
Fewer had hypertrichosis, which is hair growth beyond the head.
What’s next for VDPHL01
This data is slated to be presented at medical gatherings at some point, Waldman said.
Meanwhile, preliminary results from the second Phase 3 male trial are anticipated later this year.
Veradermics is also recruiting women for its female pattern hair loss Phase 2/3 trial.
Participants should be 18 to 65 years old with hair loss that looks like widening of the part, significantly thinning hair, greatly reduced hair volume and/or areas where hair growth has slowed or stopped completely and scalp shows through.
Participation is free, and insurance is not required.
So when could VDPHL01 hit pharmacy shelves?
“Under our current plan, if our studies continue to read out positively on the timelines that we expect, and we submit and gain FDA approval, we believe it could be within the next two to three years,” Waldman said.
Read the full article here